Goa is abuzz with excitement as vintage bike and car owners, users, collectors and fans are decking […]

ADVANCES IN CANER GENOMICS AND PRECISION ONCOLOGY! By Dr Amit Dias, MD
April 25- May 01, 2026, MIND & BODY, HEART & SOUL April 24, 2026As cancer continues to rise across the globe, so too does the science that fights it. In this special invited article, Dr Amit Dias takes readers beyond the complexity of medical jargon to familiarise them with the language — and promise — of cancer genomics and precision medicine. What once seemed like the distant future is already shaping clinical care today. This insightful piece not only explains the state-of-the-art developments transforming oncology but also reminds us that behind every innovation lies a deeper purpose: to save lives, restore dignity, and offer hope where it is needed most.
The burden of cancer: Where do we stand today?
CANCER remains one of the leading causes of morbidity and mortality worldwide, with an estimated 20 million new cases annually and nearly 10 million deaths. The burden is rising, particularly in low and middle-income countries, driven by aging populations, lifestyle transitions, environmental exposures, and improved detection.
Cancers are broadly classified into carcinomas (such as lung, breast, and colorectal cancers arising from epithelial tissues), sarcomas (from connective tissues like bone and muscle), leukemias (malignancies of blood-forming tissues), lymphomas (cancers of the lymphatic system), and central nervous system tumors involving the brain and spinal cord.
Despite advances in surgery, chemotherapy, and radiotherapy, outcomes for many cancers remain suboptimal due to late diagnosis, tumor heterogeneity, and resistance to therapy. This has driven a paradigm shift toward understanding cancer at the molecular level.

Understanding the new language of cancer care:
CANCER genomics refers to the comprehensive study of genetic alterations — such as mutations, deletions, and amplifications — in cancer cells. It aims to identify “driver mutations” responsible for tumor initiation and progression.
Oncogenomics is a specialized branch of cancer genomics focusing on genes that promote cancer, including oncogenes and tumor suppressor genes such as TP53 and BRCA1/2.
Genomic profiling involves analyzing a tumor’s genetic makeup using advanced technologies like Next-Generation Sequencing (NGS) to detect actionable mutations, thereby creating a molecular fingerprint of the tumor.
Precision medicine is a clinical approach that tailors treatment based on individual variability in genes, environment, and lifestyle, with particular emphasis on tumor genetics.
Targeted therapies are drugs designed to specifically inhibit molecular pathways essential for cancer cell survival, minimizing damage to normal cells and reducing toxicity compared to conventional therapies.
Personalized cancer treatment refers to designing therapeutic strategies uniquely suited to an individual patient based on genomic, clinical, and environmental data.
“THE future is not something we enter—it is something we create,” In the realm of cancer care, that future is unfolding now. With rapid advances in genomics, targeted therapies, and personalized treatment, medicine is moving from uncertainty to precision, from despair to hope
Recent Advances in Cancer Genomics
Cancer genomics is an evolving field driven by advances in laboratory and computational technologies, enabling a detailed examination of the molecular and cellular aspects of cancer. This field extends beyond DNA analysis to include gene expression, proteins, and other molecular features, providing a comprehensive understanding of tumor biology.
Next-generation sequencing (NGS) has revolutionized cancer research by enabling rapid, high-throughput, and cost-effective sequencing of entire genomes. It allows the identification of rare mutations, improves understanding of tumor heterogeneity, and helps detect mechanisms of drug resistance.
Liquid biopsy represents a major breakthrough as a minimally invasive technique that detects circulating tumor DNA (ctDNA) in blood. It enables early detection, monitoring of treatment response, and identification of relapse even before clinical symptoms appear.
The integration of multi-omics approaches—including genomics, transcriptomics, proteomics, and metabolomics—provides a holistic view of cancer biology, revealing complex interactions within tumor cells and their microenvironment.
Artificial intelligence and big data analytics are increasingly being used to interpret vast genomic datasets, predict treatment responses, identify novel drug targets, and enhance diagnostic precision.
Gene-editing technologies such as CRISPR are opening new frontiers by enabling precise modification of genetic sequences, with potential applications in correcting oncogenic mutations and enhancing immune-based therapies like CAR-T cells.
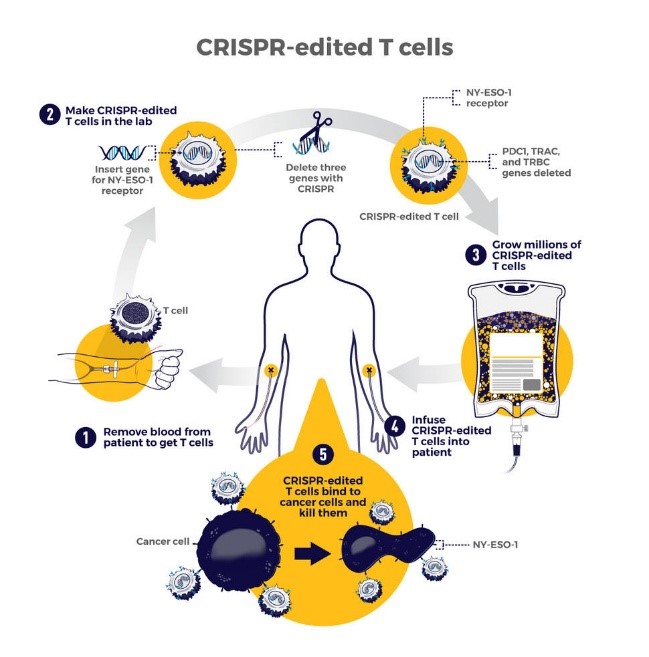
Transforming treatment: Clinical applications
THE translation of genomic discoveries into clinical practice has transformed oncology. Here are some examples.
Imatinib, a targeted therapy for chronic myeloid leukemia, inhibits the BCR-ABL fusion protein and has dramatically improved survival outcomes.
Trastuzumab is effective in HER2-positive breast cancer, significantly improving prognosis in this subgroup.
Erlotinib and Gefitinib target EGFR mutations in non-small cell lung cancer, offering effective and less toxic treatment options.
These therapies exemplify how molecular insights can lead to highly effective, tailored treatments, marking a shift from traditional “one-size-fits-all” approaches to precision oncology.
Advances in the diagnosis: Molecular classification of cancer
TRADITIONAL histopathological classification is increasingly being replaced by molecular taxonomy. Breast cancer is now classified into subtypes such as Luminal A, Luminal B, HER2-enriched, and triple-negative based on gene expression profiles. Lung cancers are categorized by mutations such as EGFR, ALK rearrangements, and KRAS mutations. Lymphomas are defined by distinct molecular signatures. This molecular classification enables more accurate diagnoses and facilitates the selection of targeted therapies based on tumor biology rather than anatomical location.
Future directions: The next frontier
THE future of cancer genomics is poised to redefine healthcare. Predictive oncology aims to identify individuals at high genetic risk and implement preventive strategies before cancer develops.
Personalized cancer vaccines, particularly mRNA-based, are being developed to target tumor-specific antigens. Nanomedicine offers targeted drug delivery systems that enhance efficacy while minimizing systemic toxicity. Pan-cancer therapies targeting specific mutations irrespective of tumor origin are also emerging, representing a shift toward mutation-based rather than site-based treatment approaches.
Ethical challenges in cancer genomics
THE rapid advancement of cancer genomics raises significant ethical challenges. Genomic data privacy is a critical concern, as such information is deeply personal and must be protected from misuse. Equity and access remain major issues, as the high cost of precision medicine risks widening disparities between populations.
Conclusion
Cancer genomics has ushered in a transformative era in oncology, shifting the focus from generalized treatment to highly individualized care. With continuous advancements in sequencing technologies, computational biology, and targeted therapies, precision medicine holds the promise of improving survival outcomes, reducing treatment-related toxicity, and potentially preventing cancer.
However, these scientific advances must be accompanied by ethical responsibility, ensuring equitable access and safeguarding patient rights. The integration of innovation with compassion and fairness will define the future of cancer care.














